|
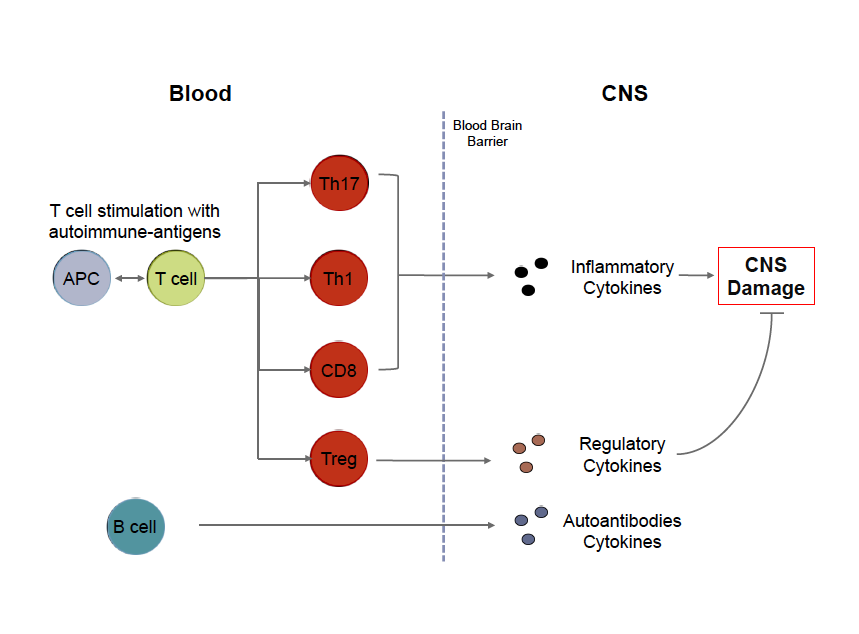
#MultipleSclerosis, #MS, #Cells, #Immunology By Brittany Goods  My goal with writing these posts is twofold. First, I'd like to present an introduction to key scientific concepts in order to help readers better understand MS research. Second I'd like to provide a resource and scientific opinion about current MS topics, ranging from therapy to basic biological research. Throughout each post, I will also try to provide links to peer reviewed open sources for readers that are interested in learning more details! Cells of MS In order to fully understand the cause of an incredibly complex disease like multiple sclerosis, it can be argued that we need to better understand the biology and hence the dysfunction of the cells that are responsible for mediating disease. It is now widely accepted that MS is an inflammatory disorder affecting the gray and white matter of the central nervous system. Whether MS is an autoimmune disease in the traditional sense, meaning that ones own immune system targets part of the body for destruction, is still a matter of considerable debate. Regardless of how this debate is resolved the cells of your immune system are involved in the pathogenesis of MS. One of the most basic questions you can ask is what role each immune cell plays in the context of MS, and further, how can we better understand what is wrong with these cells so we can fix them in a targeted way? There is a lot of technical information available to patients with MS, but they aren't often presented in a way that can be easily interpreted. I'm writing this post to give an overview of what is important about some of these key immune cells, with a focus on T cells, and hopefully provide an introduction to these without too much jargon and acronyms that inevitably complicate immunology. For those who are interested, each term in bold is linked to an open source article with a focus on MS where possible. The immune system is comprised of a vast network of specialized cells that work in concert to protect us from infection and help heal injuries. There are two arms of this system: innate and adaptive. The innate branch of cells are the first responders that are designed to sound the alarm that something is wrong. These cells are the infantry of your immune system: they're always there, ready for anything, and will report what is going on to their 'commanders' in the adaptive branch. The adaptive immune system is designed to learn about what's wrong so when the body sees the same problem they can be poised to respond quickly. In the same vein of army analogies, these cells are like the special forces. They're trained very specifically and shape the evolution of a given immune response. In the context of MS, a kind of adaptive cell called a T cell recognizes components of the CNS and initiates an immune response. Thus, T cells become activated in response to specific proteins they recognize, called an antigen. These protein antigens may be present in many different cell types in the brain although most researchers focus on protein antigens present in the myelin produced by oligodendrocytes. There are many different flavors of T cells that can arguably be broken into three main types: helper T cells, regulatory T cells, and cytotoxic T cells. Helper T cells, also called CD4 Th cells, become activated by specific antigens, such as myelin, that they recognize through specialize surface receptors. These activated cells exert their function by producing cytokines, proliferating, and expressing molecules that allow them to travel to specific sites in the body so they can activate other immune cells. In the context of MS, two types of Th cells called Th1 and Th17 cells, are implicated in disease. I will write a later post specifically focusing on these cells and what we know about them and what this might mean from a therapeutic standpoint. Regulatory T cells, also called Tregs, are needed to limit this response or turn off this response when needed. Without these cells, the signals provided by Th cells can amplify unchecked. Finally, cytotoxic T cells, also called CD8 T cells, are most important for killing cells that are infected with the antigen they recognize. These cells are kind of like the cleanup crew of an immune response. B cells are another kind of adaptive immune cell that produce antibodies that act as molecular tags to signal that the antigen they're bound to shouldn't be there. Interestingly, a few key things make the role of B cells a little more complicated: 1) B cells actually need help from T cells to begin this process, 2) B cells can activate T cells (act as what are called antigen presenting cells), and 3) B cells can modulate T cells by also secreting cytokines. For a very comprehensive review of immune cells as they pertain to MS, please check out this review. Adapted from Weiner, 2009.
B cells and T cells are the immune cells most implicated in disease based on both genetic studies and the success of treatments like those that deplete B cells (i.e.: rituximab) and prevent immune cells from trafficking to the CNS (i.e.: nataluzimab). There are still many outstanding questions about these myelin-reactive T cells and B cells as well. For example, the immune system has many checks in place to prevent the development of autoimmunity, so how is it that these cells arise? What makes these cells fundamentally different, since it has been shown that myelin-reactive T cells can be found in people that do not have MS? For B cells, how do these cells become activated to produce antibodies in the CNS? How do these cells activate T cells, which can ultimately lead to myelin-damage? Finally, how do these cells get into the CNS? My next few posts will review the science behind stem cell transplants as a therapy for MS, as well as newer genetic studies that have revealed key insights into the cells discussed above. Stay tuned and please post any questions, and future posts can address these! Other sources: "Multiple Sclerosis Immunology: A foundation for Current and Future Treatments," Takashi Yamamura and Bruno Gran. 2013. #cellsofMS #MSimmunology Brittany Goods is a doctoral candidate in Biological Engineering at the Massachusetts Institute of Technology. She received a Bachelor of Engineering degree at Dartmouth College and a Bachelor of Arts degree in Biochemistry at Colby College. She is currently an NSF graduate research fellow at MIT in Chris Love’s lab. Her research is focused in clinical immunology, with the goal of applying novel computational and experimental techniques to study multiple sclerosis. Ultimately, these efforts towards better characterization of cells responsible for initiating and propagating disease will facilitate insights into MS pathogenesis and reveal novel therapeutic targets.
Howard Parks
1/22/2017 08:29:30 pm
Ms. Goods, Comments are closed.
|
Archives
April 2015
About Dr. Debbie
Deborah Backus, PT, PhD is Director of Multiple Sclerosis Research at the Shepherd Center in Atlanta, Georgia. Dr. Debbie received her B.S. in Physical Therapy in 1986, and her Ph.D. in neuroscience in 2004. Categories
All
|
- Home
- About Us
- Virtual MS Center
- News & Resources
- Seminar Registration
- Health & Wellness
- Blogs
- About MS
-
Symptoms
- Balance and Walking Issues
- Breathing/Respiratory
- Bowel Dysfunction
- Cognitive Dysfunction
- Crying/Laughing Uncontrollably (PBA)
- Depression and Anxiety
- Dizziness/Vertigo
- Dysphagia
- Fatigue
- Foot Drop
- Hearing or Smell or Taste Changes
- Heat Sensitivity
- Leg Weakness
- Loss of Hand Dexterity and Coordination
- Memory and Mutliple Sclerosis
- Migraines
- Numbness/Tingling/Altered Sensation
- Nystagmus and Oscillopsia
- Pain
- Sexual Dysfunction
- Sleep Issues
- Spasticity/Spasms/Cramps
- Speech/Swallowing
- Urination/Bowel Problems
- Vision
- MS Clinics
- MS Topics
- Register With Us
- Terms of Use/Privacy/HIPAA
- MS HealthCare Journey

 RSS Feed
RSS Feed
